Chemistry 12 Kinetics Practice Test # 2 Iannone
1. Which of the following units could be used to express the reaction rate?
A. mL/s
B. mL/g
C. g/mL
D. mL/mol
2. Consider the reaction: Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2(g)
The rate of production of ZnCl2, can be increased by
A. decreasing the [HCl].
B. increasing
the temperature
C. increasing the volume of H2.
D. decreasing the surface area of Zn.
3. The statement, the minimum energy needed for a successful collision, defines
A. enthalpy.
B. activation
energy.
C. the ΔH of the reaction.
D. the activated complex.
4. As an activated complex changes to products,
A. potential
energy changes to kinetic energy.
B. kinetic energy changes to potential energy.
C kinetic energy changes to activation energy.
D. potential energy changes to activation energy.
5. Which of the following is most likely to have the greatest rate at room temperature.
A. 2H2(g) + O2(g) → 2H2O(l)
B. 2Ag+(aq) +
CrO42-(aq)
→ Ag2CrO4(s)
C. Pb(s) + 2HCl(aq) → PbCl2(aq) + H2(g)
D. CH4(g) + 2O2(g) → CO2(g) + H2O(g)
6 Consider the following PE diagram for an uncatalyzed and catalyzed reaction
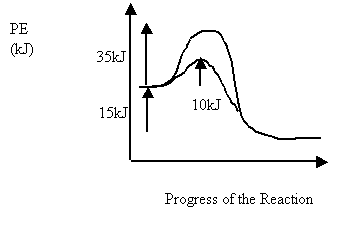
Which of the following describes the forward catalyzed reaction?
Activation Energy (kJ) ΔH (kJ)
A. 10 -15
B. 10 15
C. 25 -15
D. 25 15
7. A substance that increases the rate of a reaction without appearing in the equation for the overall reaction is a(an)
A. product
B. catalyst
C. reactant
D. intermediate
8. Activation energy can be described as the
A. energy of motion
B. energy of the activated complex.
C. energy difference between the reactants and the products.
D. energy difference
between the reactants and the activated complex.
9. What effect does a catalyst have on a reaction?
A. It changes the ΔH of a reaction.
B. It increases the kinetic energy of the reactants.
C. It decreases the potential energy of the products.
D. It provides
a reaction mechanism with a lower activation energy.
10. Consider the following reaction involving 1.0 g of powdered zinc:
Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2(g)
Trial Temperature (0C) Concentration of HCl
1 40 3.0
2 20 3.0
3 40 6.0
The rates in order of fastest to slowest are
A. 1, 2, 3
B. 2, 1, 3
C. 3, 1, 2
D. 3, 2, 1
11. Consider the following potential energy diagram for a reversible reaction:
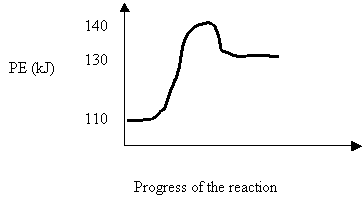
![]()
![]()
![]()
Which of the following describes the system above?
Reaction Activation Energy (kJ) ΔH (kJ)
A. reverse 10 -20
B. reverse 10 -30
C. forward 30 +10
D. forward 20 +30
12. An activated complex is a chemical species that is
A. stable and has low PE.
B. stable and has high PE.
C. unstable and has low PE.
D. unstable and has high PE.
13. Consider the reaction: Ca(s) + 2H2O(l) → Ca(OH)2(aq) + H2(g)
At a certain temperature, 2.05 g Ca reacts completely in 30.0 seconds. The rate of consumption of Ca is
A. 0.00208 mol/min
B. 0.0833 mol/min
C. 0.102
mol/min
D. 5.00 mol/min
14. Increasing the temperature of a reaction increases the reaction rate by
I. increasing frequency of collision
II. increasing the kinetic energy of collision
III. decreasing the potential energy of the collision
A. I only.
B. I and II
only.
C. II and III only.
D. I, II, and III.
15. A certain reaction is able to proceed by various mechanisms. Each mechanism has a different Ea and results in a different overall rate. Which of the following best describes the relationship between the Ea values and the rates?
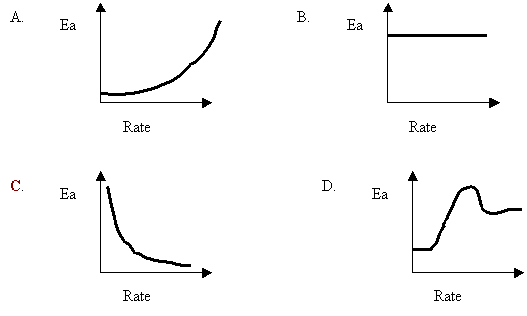
16. For collisions to be successful, reactants must have
A. favourable geometry.
B sufficient heat of reaction only.
C. sufficient potential energy only.
D. sufficient
kinetic energy and favourable geometry.
17. Consider the following reaction: 1/2 H2(g) + 1/2 I2(g) → HI(g) ΔH = +28 kJ
The activation energy for the formation of HI is 167 kJ. The activation energy for the decomposition of HI is
A. 28 kJ
B. 139 kJ
C. 167 kJ
D. 195 kJ
18. Which of the following factors affects the rate of heterogeneous reactions only?
A. nature of the reactant
B. temperature
C. surface
area of reactants
D. concentration of reactants
19. A 25.0 mL sample of hydrogen peroxide decomposes producing 50.0 mL of oxygen gas in 137 s. The rate of formation of O2 in mL/min is
A. 0.182 mL/min
B. 0.365 mL/min
C. 10.9 mL/min
D. 21.9
mL/min
20. Consider the following reaction mechanism:
step 1 2NO + H2 → N2 + H2O2
step 2 H2O2 + H2 → 2H2O
In this reaction H2 is a
A. product
B. catalyst
C. reactant
D. reaction intermediate
21. Which of the following properties could be used to measure the rate of the following reaction taking place in an open container?
Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2(g)
A. mass of Zn
B. solubility of HCl
C. concentration of Cl-
D. colour of the solution
22. Consider the following reaction: N2 + 3H2 → 2NH3
The rate of formation of NH3 is 3.0 mole/min. The rate of consumption of H2 is:
A. 1.5 mole/min
B. 2.0 mole/min
C. 4.5
mole/min
D. 9.0 mole/min
23. Consider the following reaction mechanism:
Step 1 NO2 + NO2 → N2O4
Step 2 N2O4 + CO → CO2 + NO + NO2
In the overall reaction, N2O4 is a
A. product
B. catalyst
C. reactant
D. reaction
intermediate
24. Consider the following mechanism:
Step 1 NO + O3 → NO2 + O2
Step 2 O + NO2 → NO + O2
The catalyst is
A. O2
B. O3
C. NO
D. NO2
25. Consider the following reaction: Mg(s) + 2HCl(aq) → MgCl2(aq) + H2(g)
The rate of this reaction increases when more Mg is added. This change is caused by the
A. addition of a catalyst
B. increase
in surface area
C. change in the nature of the reactants
D. increase in concentration of reactants
26. Consider the following PE diagram
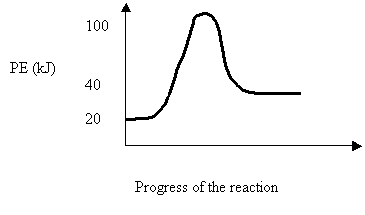
The forward reaction can be described as
DH Ea Type
A. +20 80 endothermic
B. +20 60 exothermic
C. -20 80 exothermic
D. -20 100 endothermic
27. Consider the following reaction: HgO(s) → Hg(l) + 1/2O2(g)
The rate of this reaction can be expressed as
A. rate = [O2}1/2
B. rate =
Δ[O2]/Δt
C. rate = Δ[Hg]/Δt
D. rate = Δ[HgO]/Δt
28. Which of the following would react most rapidly?
A. Powdered Zn in 1.0 M HCl at 25 0C
B. Powdered Zn in 20 M HCl at 40 0C
C. A lump of Zn in 2.0 M HCl at 25 0C
D. A lump of Zn in 1.0 M HCl at 40 0C
29. Addition of a catalyst to a reaction increases the rate because it
A. increases the value of ΔH
B. decreases the value of ΔH
C. provides an alternate mechanism with a lower Ea
D. provides an alternate mechanism with a higher Ea
30. When a collision occurs between two reactant species which possess between them the minimum kinetic energy a product does not always form. This may be a result of
A. low temperature
B. small surface area
C. low concentrations
D. unfavourable
geometry
Subjective Answers
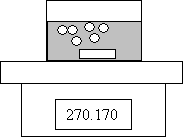
1. An experiment is done to determine the rate of the following reaction:
 2Al(s) +
6HCl(aq) → 3H2(g)
+ 2AlCl3(aq)
2Al(s) +
6HCl(aq) → 3H2(g)
+ 2AlCl3(aq)
The following data are collected
Time (s) Mass of Flask + Contents (g)
0.0 270.230
30.0 270.200
60.0 270.170
60.0 s 0.060 g H2
Calculate the rate of consumption of Al in mol/min. (3 marks)
0.060 g H2 x 60 s x 1mole
H2 x 2 mol Al
![]()
60.0 s 1 min 2.0
g 3 mol H2
= 0.020 mol Al/min
2. Define the term activation energy.
The minimum energy required for a successful collision.
3. Define the word Activated complex.
Unstable
reaction intermediate with high potential energy and low kinetic energy.
4. Define the word mechanism.
A sequence of steps that determines the overall reaction.
5. Consider the following reaction mechanism
Step 1 ?
Step 2 H2 + Cl → HCl + H
Step 3 H + Cl2 → HCl + Cl
Step 4 Cl + Cl → Cl2
Overall H2 + Cl2 → 2HCl
a) Write the equation for step 1
Cl2 → 2Cl
b) Identify the reaction intermediate(s)
Cl H
6. Consider the overall reaction: 4HBr + O2 → 2H2O + 2Br2
A proposed three-step mechanism is:
Step 1 HBr + O2 → HOOBr
Step 2 ?
Step 3 HBr + HOBr → H2O + Br2
Write the equation for step 2.
2HBr + HOOBr
→ HOBr + H2O + Br2
7. A student wishes to monitor the rate of the following reaction:
CaCO3(s) + 2HCl(aq) → CaCl2(aq) + CO2(g) + H2O(l)
Identify two properties that could be used to monitor the rate of the reaction. Describe and explain the changes that would occur.
Property 1 Mass of CaCO3(s) over time
Change and explanation Decreases, as reactants are converted into products.
Property 2 Concentration of HCl over time
Change and explanation Decreases, as reactants are converted into products.
Any
two in this list! Concentration of CaCl2(aq)
Increases,
as reactants are converted into products
Volume of CO2
Increases,
as reactants are converted into products
Mass of
an open beaker
Decreases
as CO2 escapes
Pressure
of a closed system over time
Increases as CO2 is produced over time
8. Carbon burns in air according to the following equation:
C(s) + O2(g) → CO2(g)
List four ways the rate of the above reaction could be increased.
Increase
temperature Increase
O2 concentration
Increase
the pressure Add a
catalyst
Increase C surface area
9. Sketch the potential energy diagram for an endothermic reaction in the space below. On your diagram clearly label:
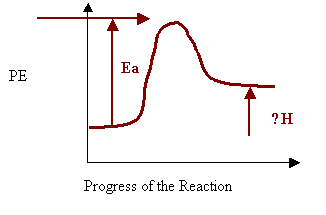
i) the energy of the activated complex
ii) the activation energy
iii) ΔH